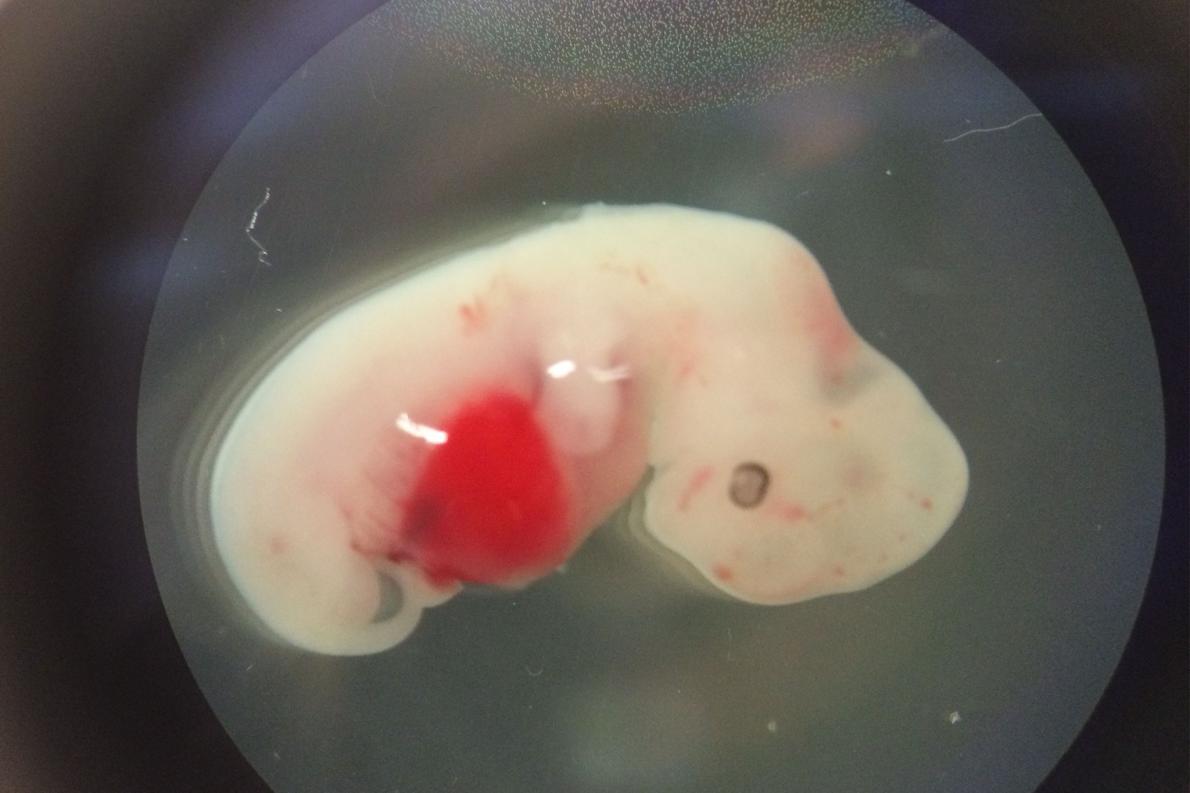
Scientists announced this week that they have created the first successful human-animal hybrid. The project now establishes that human cells can be introduced into a non-human organism (in this case a pig), survive, and grow inside a host animal. This advance in science has been a long time coming and a dream of many. Because someone is added every ten minutes to the national waiting list for organ transplants, this new discovery might help to ease our critical shortage of donor organs.
An international team of researchers, led by the Salk Institute, published their findings in the journal Cell, after having created what’s scientifically known as a chimera: an organism that contains cells from two different species. Though experiments like this are ineligible for public funding in the United States, the team was able to work thanks to private donors.
Beginning with embryos, one animal’s cells were introduced into the embryo of another and then allowed to grow together into a hybrid. While it may sound odd and science fiction-y, “it’s a way to eventually solve a number of vexing biological problems with lab-grown organs,” reports the article. The issue arises when you try to convince stem cells to grow into the specific tissues and organs needed.
So, using concepts already figured out by other scientists, Juan Carlos Izpisua Belmonte, a professor in the Salk Institute’s Gene Expression Laboratory and his team:
“…using the genome editing tool called CRISPR to hack into mouse blastocysts—the precursors of embryos. There, they deleted genes that mice need to grow certain organs. When they introduced rat stem cells capable of producing those organs, those cells flourished.
The mice that resulted managed to live into adulthood. Some even grew gall bladders, which haven’t been part of the species for 18 million years.”
Risk of Rejection
When the team took stem cells from rats and injected them into pig blastocysts, it failed, likely because rats and pigs have dramatically different gestation times and evolutionary ancestors. However, pigs have similarity to humans and although they take less time to gestate, their organs look a lot like ours. But the team discovered that still when introducing human cells into the pigs without killing them, timing was everything.
More from the article:
“We tried three different types of human cells, essentially representing three different times” in the developmental process, explains Jun Wu, a Salk Institute scientist and the paper’s first author. Through trial and error, they learned that naïve pluripotent cells—stem cells with unlimited potential—didn’t survive as well as ones that had developed a bit more.
When those just-right human cells were injected into the pig embryos, the embryos survived. Then they were put into adult pigs, which carried the embryos for between three and four weeks before they were removed and analyzed.
In all, the team was able to create 186 later-stage chimeric embryos that survived and “we estimate [each had] about one in 100,000 human cells,” said Wu.
Currently, the embryos contain too much pig tissue to be tolerated by humans- but they are at least over the first big hurdle. The next big step is to attempt to increase the number of human cells.
More study needs to be done but the team is excited about the breakthrough.